Traditional methods for enzyme production often rely on cultivating microorganisms in large-scale fermenters. These methods can be time-consuming and resource-intensive, often yielding enzymes with limited activity or stability. The natural variability inherent in these biological systems can lead to unpredictable results and difficulties in optimizing enzyme properties for industrial applications. Moreover, these methods typically involve a lengthy screening process to identify suitable strains with desired traits.
The selection and optimization of existing microorganisms to produce enzymes has historically been a trial-and-error process. This approach can be inefficient and costly in terms of time and resources. The ability to directly manipulate the genetic material of the organism offers a more precise and efficient alternative.
Harnessing Synthetic Biology for Enhanced Enzyme Production
Synthetic biology offers a powerful toolkit for enhancing enzyme production. By introducing specific genetic modifications, researchers can fine-tune the expression levels of enzyme-encoding genes, leading to higher yields of the desired enzyme. This approach allows for the targeted optimization of enzyme properties such as activity, stability, and selectivity.
Furthermore, synthetic biology allows for the introduction of novel regulatory mechanisms. This precise control over enzyme production enables researchers to achieve a more efficient and predictable production process. The ability to engineer organisms for optimal enzyme production in a controlled environment is a significant advancement over traditional methods.
Future Applications and Challenges in Enzyme Production
The application of synthetic biology in enzyme production promises significant advancements in various industries. From biofuel production to pharmaceuticals, enzymes play a crucial role, and synthetic biology allows for the creation of enzymes with tailored properties to meet specific needs. This could lead to more sustainable and efficient industrial processes.
Despite the immense potential, challenges remain in the field. Ensuring the safety and ethical implications of genetically modified organisms (GMOs) is a critical concern. Furthermore, the development of robust and scalable synthetic biology platforms for enzyme production requires further research and investment. Overcoming these challenges will be crucial for realizing the full potential of synthetic biology in enzyme production.
Enzyme Engineering for Enhanced Functionality
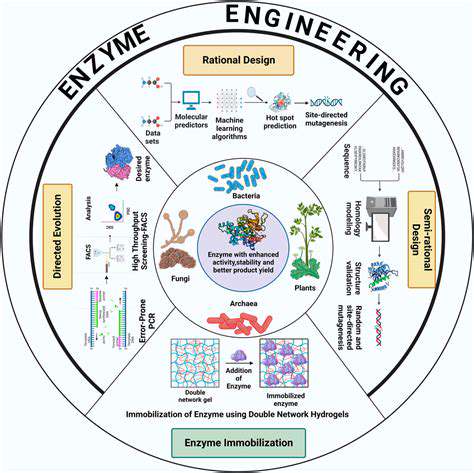
Enzyme Engineering Strategies for Improved Functionality
Enzyme engineering, a powerful tool in biotechnology, focuses on modifying existing enzymes or creating entirely new ones with enhanced characteristics. This process involves altering the amino acid sequence of the enzyme, often through site-directed mutagenesis, to modify its structure and consequently its function. This targeted approach allows for the optimization of enzymes for specific industrial or biotechnological applications. Such improvements can include increased stability, improved substrate specificity, and enhanced catalytic efficiency.
Rational Design and Directed Evolution
Rational design utilizes our understanding of enzyme structure and function to predict and implement changes in the amino acid sequence. This approach leverages computational modeling and structural biology to guide the design process. However, it is often limited by our incomplete understanding of complex enzyme-substrate interactions.
Directed evolution, on the other hand, uses iterative rounds of mutagenesis and selection to evolve enzymes with improved properties. This process mimics natural evolution, allowing enzymes to adapt to specific conditions. This method is often used for complex systems where rational design is less effective.
Protein Engineering Techniques
A variety of protein engineering techniques are employed in enzyme engineering. Site-directed mutagenesis is a common approach, enabling precise modifications of specific amino acid residues. Other techniques include error-prone PCR, random mutagenesis, and DNA shuffling, which introduce random mutations in the enzyme's DNA sequence. Each method has its own advantages and limitations, and the choice of technique depends on the specific enzyme and desired improvement.
Improving Enzyme Stability
Enzymes often lose activity when exposed to extreme temperatures, pH values, or organic solvents. Enzyme engineering can significantly improve their stability under these challenging conditions. This is crucial for industrial applications where enzymes operate under harsh environments. Improving stability also reduces the need for expensive and energy-intensive enzyme replacement during industrial processes.
Enhancing Catalytic Efficiency
Improving the catalytic efficiency of enzymes is a significant goal in enzyme engineering. This involves optimizing the enzyme's active site to enhance the rate of substrate conversion. Several factors contribute to this process, such as optimizing substrate binding, reducing the activation energy, and improving the overall catalytic mechanism.
Targeting Specific Applications
Enzyme engineering is not a one-size-fits-all approach. Researchers frequently tailor the engineering strategies to meet the specific needs of a given application. For instance, enzymes used in food processing may need to exhibit enhanced stability at high temperatures, whereas those in biofuel production might require enhanced substrate specificity for particular sugars. Focusing on specific applications allows for the development of tailored enzymes with optimal properties for that unique process.
The Future of Enzyme Engineering
The field of enzyme engineering is constantly evolving, with new tools and techniques emerging that enhance our ability to manipulate enzymes. Advancements in computational modeling, high-throughput screening, and synthetic biology are paving the way for even more sophisticated enzyme engineering strategies. This will lead to more efficient and sustainable industrial processes, and will also open new avenues for addressing global challenges in medicine, agriculture, and environmental remediation. The future promises a deeper understanding of enzyme function and the creation of enzymes optimized for specific tasks.
Boosting Enzyme Production Through Metabolic Engineering
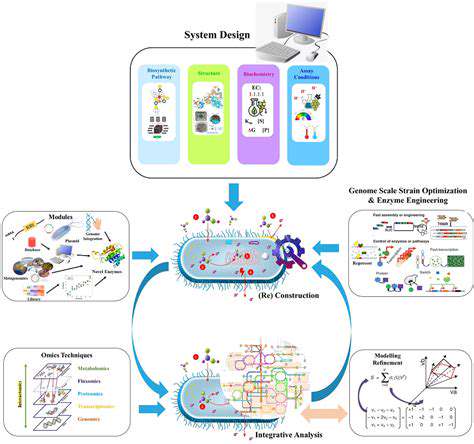
Optimizing Growth Conditions
Enzymes are biological catalysts that significantly accelerate biochemical reactions within living organisms. To maximize enzyme production, it's crucial to understand and optimize the growth conditions of the host organism. Factors like temperature, pH, and nutrient availability play a critical role in influencing enzyme synthesis. Finding the optimal balance for these factors is paramount for achieving high yields of the desired enzyme.
Maintaining consistent and controlled environments is vital. Fluctuations in these parameters can lead to suboptimal enzyme production. Precise control over these conditions through careful monitoring and adjustment is essential for achieving a robust and reliable enzyme production process.
Employing Genetic Engineering Techniques
Genetic engineering offers powerful tools to manipulate the genetic makeup of organisms, thereby enhancing enzyme production. Techniques like gene cloning, overexpression, and the introduction of specific promoters can be leveraged to increase the expression levels of the desired enzyme gene. This approach allows for a targeted and often considerable boost in enzyme production.
Furthermore, the introduction of specific regulatory elements, such as promoters and enhancers, can significantly impact the efficiency of gene expression. These modifications can fine-tune the production rate to match specific needs or environmental conditions.
Harnessing Metabolic Engineering Strategies
Metabolic engineering strategies aim to re-route cellular metabolism to prioritize the production of the desired enzyme. This involves manipulating the metabolic pathways that lead to the enzyme's synthesis. By strategically altering the expression of key metabolic enzymes, the organism can be directed towards enhanced enzyme production. This approach often involves a detailed understanding of the organism's metabolic network.
The identification and manipulation of key metabolic intermediates or pathways can lead to substantial improvements in enzyme yields.
Utilizing Microbial Systems
Microorganisms, such as bacteria and fungi, are commonly used for enzyme production due to their fast growth rates and ease of genetic manipulation. Their ability to rapidly multiply in controlled environments makes them ideal candidates for high-throughput enzyme production. Specific strains of microorganisms, engineered to possess enhanced enzyme production capabilities, can be employed to maximize yields.
The selection of appropriate microbial strains is crucial in this process. Factors such as growth rate, tolerance to stress conditions, and ease of cultivation significantly impact the overall efficiency of enzyme production.
Implementing Bioreactor Optimization
Bioreactors are essential tools for large-scale enzyme production. Optimizing bioreactor conditions, including aeration, mixing, and temperature control, is critical for efficient enzyme synthesis. Proper aeration ensures adequate oxygen supply for the microorganisms, while optimal mixing maintains uniform conditions throughout the culture. Precise control of temperature ensures optimal enzyme activity.
The design and operational parameters of the bioreactor significantly impact the overall yield and quality of the produced enzyme. Careful consideration of these factors leads to a more efficient and cost-effective production process.
Exploring Immobilization Techniques
Enzyme immobilization is a technique that can enhance enzyme stability and reusability. This involves attaching the enzyme to a solid support material, such as beads or membranes. This approach protects the enzyme from degradation and allows for repeated use, reducing costs and improving efficiency in industrial applications. Various immobilization methods exist, each with its advantages and disadvantages.
Immobilized enzymes often exhibit enhanced stability compared to free enzymes, which is a significant advantage in industrial settings where repeated use is desired.
Considering Cost-Effectiveness
A crucial aspect of enzyme production is minimizing the overall cost. This involves optimizing the entire process, from raw material procurement to bioreactor operation. Careful selection of feedstocks and optimization of culture conditions can dramatically reduce production costs. The use of readily available and affordable substrates is also a key consideration. Waste minimization and efficient resource utilization are equally essential for cost-effective enzyme production.
Implementing strategies to reduce energy consumption and maximize resource utilization will contribute significantly to lowering the overall cost of enzyme production. This is crucial for making enzyme production commercially viable.